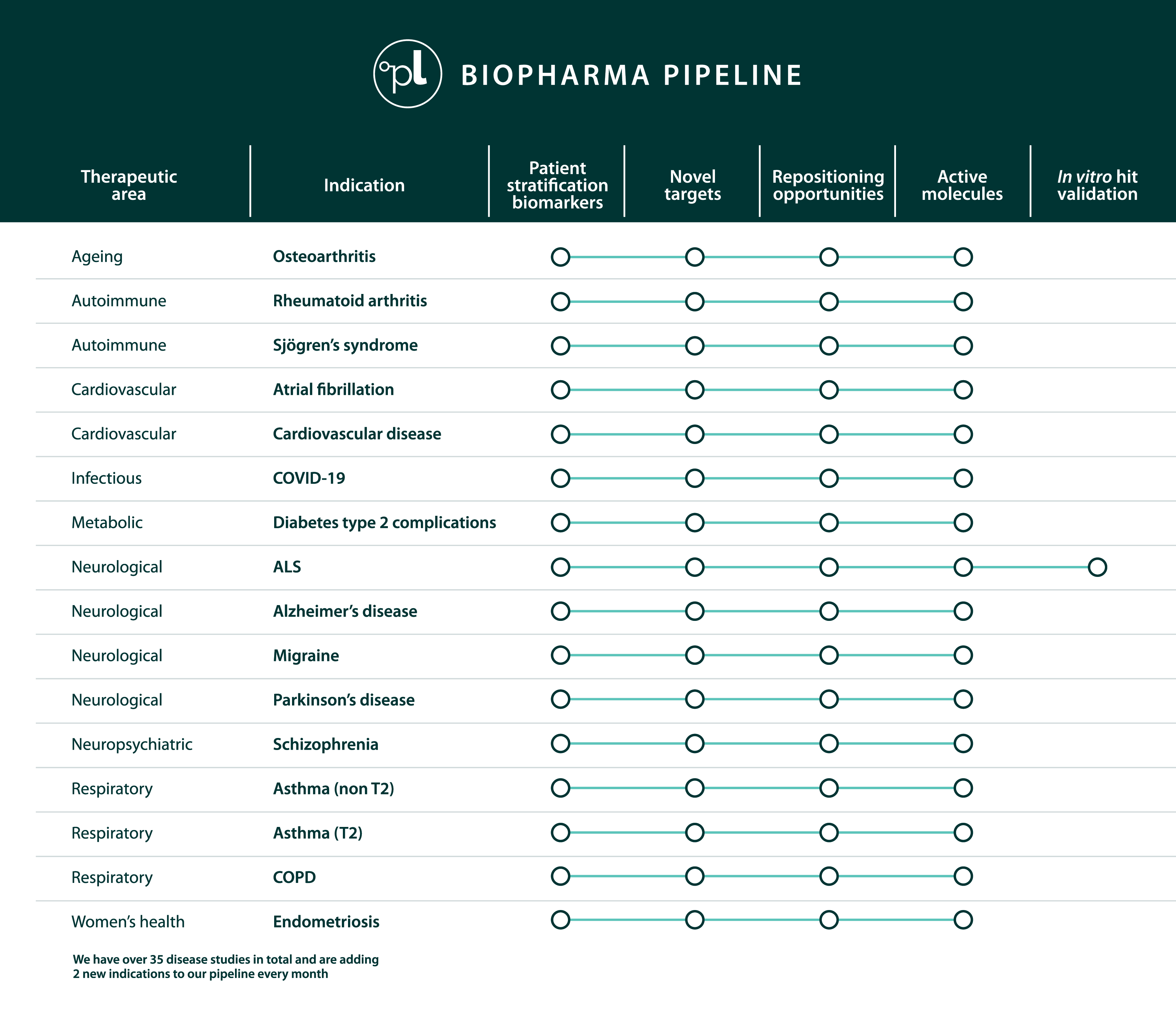
Our pipeline
One of the largest drug discovery pipelines of precision targeted therapies for unmet medical needs
Chronic diseases
analyzed
Novel drug targets
discovered
Indication extensions
identified
Chronic diseases
analyzed
Novel drug targets
discovered
Indication extensions
identified
Drug discovery pipeline
Our pipeline includes novel drug targets with genetic linkage and indication expansion opportunities at all stages of development - all with highly specific patient stratification biomarkers.
See more
See less
High-resolution patient stratification results:
- 520 Disease risk loci
- 250 Risk associated genes mapped to disease associated mechanisms
- >30 Druggable targets
- 57 Known active compounds
See more
See less
High-resolution patient stratification results:
- 717 Disease risk loci
- 299 Risk associated genes mapped to disease associated mechanisms
- >30 Druggable targets
- 263 Known active compounds
See more
See less
High-resolution patient stratification results:
- 158 Disease risk loci
- 88 Risk associated genes mapped to disease associated mechanisms
- 23 Druggable targets
- 59 Known active compounds

See more
See less
High-resolution patient stratification results:
- 35 Disease risk loci
- 20 Risk associated genes mapped to disease associated mechanisms
- 8 Druggable targets
- 120 Known active compounds
See more
See less
High-resolution patient stratification results:
- 73 Disease risk loci
- 33 Risk associated genes mapped to disease associated mechanisms
- 16 Druggable targets
- 19 Known active compounds

See more
See less
High-resolution patient stratification results:
- 267 Disease risk loci
- 113 Risk associated genes mapped to disease associated mechanisms
- >30 Druggable targets
- 732 Known active compounds

See more
See less
High-resolution patient stratification results:
- 707 Disease risk loci
- 341 Risk associated genes mapped to disease associated mechanisms
- >30 Druggable targets
- 2611 Known active compounds

See more
See less
High-resolution patient stratification results:
- 125 Disease risk loci
- 85 Risk associated genes mapped to disease associated mechanisms
- >30 Druggable targets
- 155 Known active compounds

(non-T2)
See more
See less
High-resolution patient stratification results:
- 570 Disease risk loci
- >200 Risk associated genes mapped to disease associated mechanisms
- >30 Druggable targets
- 281 Known active compounds

(T2-high)
See more
See less
High-resolution patient stratification results:
- 359 Disease risk loci
- 163 Risk associated genes mapped to disease associated mechanisms
- >30 Druggable targets
- >300 Known active compounds

See more
See less
High-resolution patient stratification results:
- 233 Disease risk loci
- 136 Risk associated genes mapped to disease associated mechanisms
- >30 Druggable targets
- 74 Known active compounds

Last updated May 24, 2023
Indication extensions
We have identified hundreds of indication extension opportunities across all stages of development. These licensable assets have high market value and strong efficacy potential for repositioning into new disease areas using patient stratification biomarkers to match them to clinically relevant patient subgroups.
Last updated March 30, 2023
Upcoming innovation
We are running new disease studies all the time, adding two new chronic disease indications to our DiseaseBank every month. Contact us to talk about your priorities and discuss partnering opportunities in one of our current or upcoming disease studies.
Partner with us-
Alopecia
-
Vitiligo
-
Age-related macular degeneration
-
Biopolar disorder
-
Chronic fatigue syndrome / ME
- Inflammatory bowel disease
- Interstitial pulmonary fibrosis
- Asthma COPD overlap syndrome
- Uterine fibroids
- Prostate cancer
- Glaucoma
Please view our pipelines on a larger screen or device for the best experience.