PRESS RELEASE
PrecisionLife and Ovation identify first potential genetic biomarkers to quantitatively predict GLP-1 efficacy response
Building on strong efficacy results, the companies will expand their collaboration to 25,000 patients to identify safety, tolerability, and drug-specific response signals
OXFORD, UK and PORTLAND, ME – 25 February 2026 – Ovation.io, a leading human omics and clinical data company, and PrecisionLife, a precision medicine company transforming how complex chronic diseases are understood and managed, today announced the results of the first phase of their collaboration to develop drug-response biomarkers to quantitively predict the efficacy, safety and tolerability of glucagon-like peptide-1 receptor agonist (GLP-1) therapies, and the extension of their partnership.
Using Ovation’s longitudinal clinical and omics datasets and PrecisionLife’s AI-driven combinatorial analytics platform, the partners identified underlying genetic drivers of GLP-1 efficacy to predict and stratify strong and weak responders within a large real-world population. The insights provide a better understanding of the genetic and biological drivers of GLP-1 responses and provide new biomarkers for drug development, refined patient enrollment in clinical trials, and potentially underpin a payor-facing test to inform reimbursement policy for these widely prescribed drugs based on a patient’s potential to tolerate and respond to a specific therapy.
GLP-1 therapies are among the fastest-growing drug classes globally, yet approximately 50% of patients discontinue treatment within 12 months, often losing previously achieved metabolic benefit[1]. With US spending on GLP-1 medicines exceeding $70 billion annually[2], variable response and high discontinuation rates create significant clinical and economic challenges for payors, providers, and pharmaceutical developers.
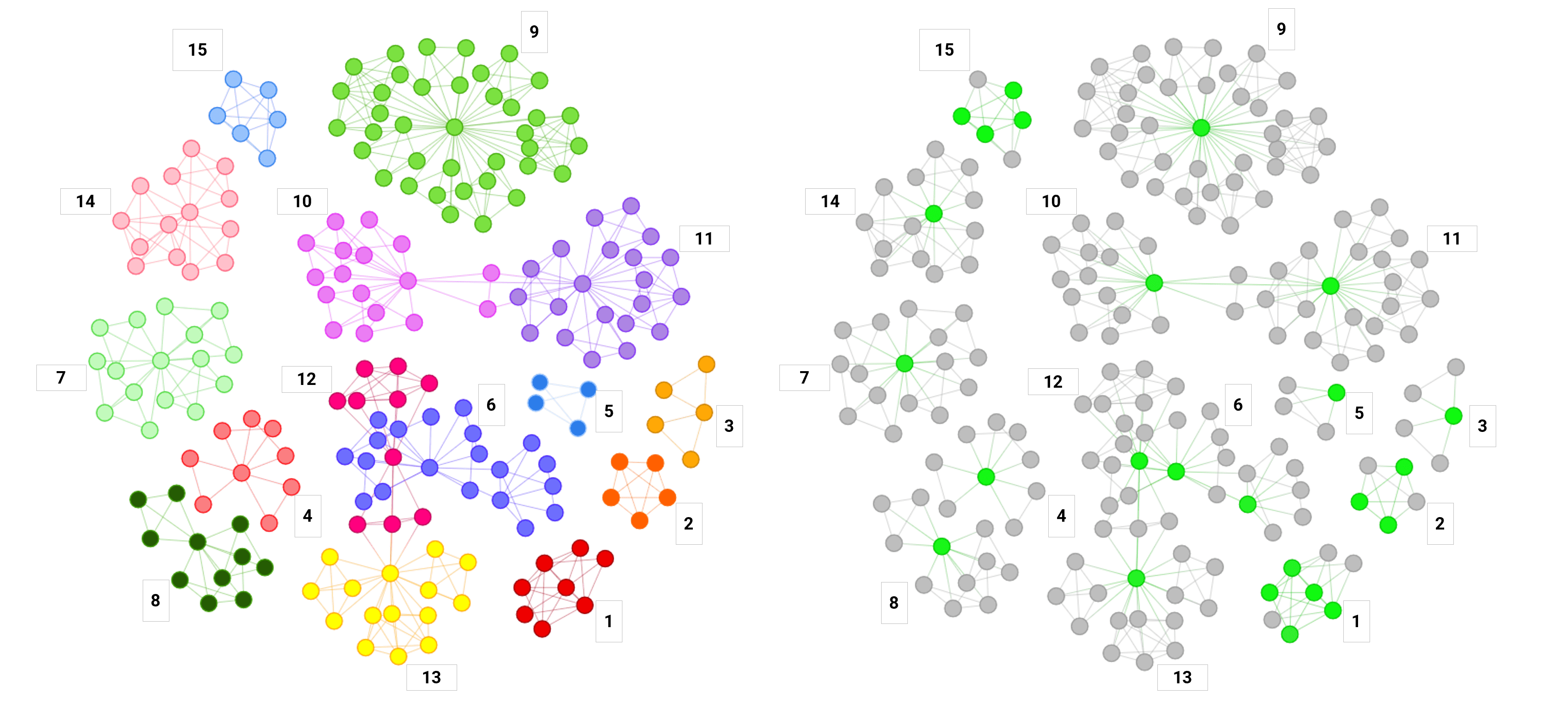
Phase 1 of the collaboration, which was initiated in December 2025, utilized 4,600 patients’ data and generated over 2,500 genetic signatures associated with GLP-1 RA-mediated efficacy. These signatures were mapped to 1,100 genes with 15 main genetic mechanisms driving efficacy. These markers of strong responses to GLP-1 drugs were also associated with type-II diabetes, cardiovascular disease, lipid metabolism and other disease states. The biomarkers of efficacy were identified in 100% of the patients.
Steve Gardner, Chief Executive Officer of PrecisionLife, stated: “While questions have been raised in the literature about the impact of genetics on GLP-1 patient responses, Phase 1 of the PrecisionLife/Ovation collaboration demonstrated that we can identify genetic biomarkers associated with strong and weak responders to GLP-1 RA drugs and quantitatively predict the level of efficacy based on their degree of response measured by BMI and HbA1c changes. While we see many known pathways, a number of the genes identified are outside of the “defined” GLP-1RA mechanisms of action and novel in the literature. We believe this initial effort demonstrates our combined ability to provide solutions in high value disease states where precision medicine and therapy selection improve outcomes and address unmet needs.”
Based on these results, PrecisionLife and Ovation have agreed to expand their partnership into Phase 2, scaling the dataset to up to 25,000 patients and incorporate more detailed clinical phenotype data. This next phase will validate and refine efficacy signals and identify predictive markers of safety and tolerability, including drug-specific response differences. This will stratify different subgroups within the diverse treatment population, showing which are most likely to be responsive to GLP-1s and which are not, and which have the highest likelihood of specific adverse events.
“Our initial, hypothesis free results suggest that by combining Ovation’s unique normalized and standardized longitudinal data sets and clinical records with PrecisionLife’s mechanistic patient stratification analysis platform, we can identify specific combinations of genetic and biological drivers that create different “response-defined” subgroups within this crucial therapeutic category,” said Curt Medeiros, Chief Executive Officer of Ovation.io. “These real-world, in-the-wild, data can be of significant value to our biopharma clients as well providing important clinically facing markers that improve patient outcomes. Matching patients to specific therapies based on their individual risk and response mechanisms, rather than treating them as a homogeneous population, enables more targeted, de-risked development programs and more accurate patient prescriptions.”
[1] Gleason et al., JMCP (2024)
[2] Tsipas et al., JAMA Netw Open (2025)
-- ENDS --
Frequently Asked Questions
GLP-1 drugs are among the fastest-growing drug classes globally, yet more than 50% of patients discontinue within 12 months. When patients stop therapy, many rapidly lose the metabolic and cardiovascular benefits they gained.
In the US alone, approximately $72 billion was spent last year on GLP-1 therapies. High discontinuation and variable efficacy mean a substantial proportion of that spend does not translate into sustained health benefit.
A predictive test could help ensure the right patients receive the right therapy that they will find safe, efficacious and tolerable, improving outcomes while reducing clinical and financial waste.
Phase 1 analyzed whole genome and limited clinical data from 4,600 GLP-1 RA patients and demonstrated that we can:
- Identify strong and weak responders based on genetic signatures
- Quantitatively predict efficacy using BMI and HbA1c change
- Detect over 2,500 genetic signatures mapping to 1,100 genes
- Identify 15 major biological mechanisms driving response
This is the first time detailed mechanistic efficacy signals have been shown directly within a GLP-1-treated real-world population at this level of resolution.
Most existing tests rely on small gene panels derived from GWAS or known appetite-related genes.
This collaboration analyzes quantitative drug response directly in GLP-1-treated patients, identifying a large number of combinatorial genetic patterns across multiple biological pathways and tissues.
Rather than testing a handful of well-known genes, the platform uncovers much deeper mechanism-level drivers of response and non-response, including multiple novel pathways beyond classical GLP-1 biology. As well as efficacy for weight loss, glycemic control and other phenotypes, we are also examining the safety and tolerability of these drugs.
We can also use this to develop drug specific tests, looking at the relative performance and benefits of one compound over another for specific patients.
The many pathways identified include:
- Dopaminergic signaling
- Glutamatergic signaling
- Leptin-mediated pathways
- PI3K-AKT signaling
- Histamine signaling
- Insulin secretion and lipid metabolism
These mechanisms influence appetite regulation, reward processing, energy balance, glucose homeostasis, and metabolic control. Importantly, some identified genes fall outside established GLP-1 mechanisms, suggesting novel biology.
By identifying biological mechanisms rather than isolated genes, the analysis helps explain why some patients experience strong, durable benefit while others do not, supports quantitative prediction of response magnitude and tolerability, and provides a mechanistic foundation for more precise prescribing and drug-specific stratification as the GLP-1 class continues to expand.
Phase 2 expands the dataset to up to 25,000 patients and bring in more detailed longitudinal clinical data.
It will:
- Validate the efficacy signals at larger scale, and at higher resolution
- Identify predictive markers of safety and tolerability
- Assess drug-specific differences in different applications
- Evaluate discontinuation and rebound risk
- Enable head-to-head drug comparisons as data matures
This moves the program from proof-of-concept to clinically actionable stratification.
Payors face significant losses associated with GLP-1 coverage. Large US insurers have reported hundreds of millions in annual losses.
A predictive test could:
- Reduce ineffective prescriptions and improve the affordability of healthcare
- Lower tolerability issues (side-effects) and discontinuation rates
- Support sustainable expansion of coverage to patients who will benefit long-term
- Enable differentiated reimbursement strategies for their health plans
This shifts GLP-1 prescribing from trial-and-error to evidence-guided allocation.
Biopharma companies face:
- Massive competition and potential market saturation with over 500 GLP-1-related clinical trials across 100+ indications
- Downward price pressure
- Highly restricted reimbursement and resistance to new indications
Mechanism-based stratification can:
- Enrich clinical trials for responders (response biomarkers)
- Identify strong responder subgroups (complementary diagnostic)
- Differentiate competing assets
- Support development in new indications (biomarker-led indication switching)
- De-risk late-stage programs (clinical trial design)
Previous efforts to expand GLP-1s into new indications - such as the semaglutide Alzheimer’s trial - largely targeted broad patient groups defined by their clinical symptoms or diagnostic labels. These approaches do not account for the fact that complex chronic diseases are driven by multiple distinct biological mechanisms, and that patients have different genetic and comorbidity backgrounds meaning they will respond differently to modulation of GLP-1 pathways.
PrecisionLife’s platform finds more pathways and applies mechanistic patient stratification, identifying the specific combinations of genetic and biological drivers that create different subgroups within a disease. This allows us to compare the broader range of biology to find true candidates for GLP-1 approaches.
This reveals which subpopulations in new indications are most likely to be responsive to GLP-1s and which are not, and which have the highest likelihood of specific adverse events. By matching therapies to the biological mechanisms active in each subgroup - rather than treating all patients as a homogeneous population - PrecisionLife enables far more targeted, de-risked development in secondary indications.
This stratified approach avoids the one-size-fits-all assumptions that have constrained past indication extension attempts and provides a clearer, evidence-led path to expanding GLP-1 use where it is biologically and clinically justified.
About PrecisionLife®
PrecisionLife is transforming how chronic diseases are predicted, treated, and prevented. Using its proprietary combinatorial analytics AI platform, PrecisionLife reveals more insights from less data than any other form of genetic analysis and uniquely discovers the biological mechanisms that drive complex diseases.
OThis approach enables high-resolution patient stratification and precision medicine at scale, powering world-leading personalized health solutions across healthcare and life sciences..
PrecisionLife partners with healthcare providers, payors and biopharma innovators to deliver clinically actionable insights that improve health outcomes, reduce costs, and extend healthy lifespan for billions of people affected by chronic disease.
For more information:
Visit: precisionlife.com
Contact: press@precisionlife.com
Follow: PrecisionLifeAI
About Ovation
Ovation is a genomic data company committed to unlocking the potential of human genomic data at scale and accelerating precision medicine development. By providing access to high-quality genomic data linked to rich, longitudinal phenotypic data at scale, Ovation allows life sciences researchers to advance drug discovery and development more efficiently. Ovation’s cloud-based LIMS and Ovation Research Network, enable a diverse network of clinical laboratories to quickly adopt innovative molecular tests while biobanking and transforming samples to be used for research.
For more information about Ovation, visit www.ovation.io
Name Surname, Position, Company



.png?width=1920&height=500&name=Predicting%20GLP-1%20Response%20(1).png)