
Unraveling complex disease biology
Chronic diseases affect billions of people and account for the majority of global healthcare costs, yet their complex, multifactorial biology makes them the hardest conditions to diagnose and treat – resulting in major unmet medical needs.
Our platform is the first to reveal the complex interplay of multiple genes and other heterogenous factors underpinning disease biology, to discover the specific causes of chronic diseases.
PrecisionLife's unique approach enables the stratification of patient populations at unprecedented resolution, accurate diagnosis and triage of patients and the linking of individuals to effective treatments by the mechanisms of their disease.
World-leading precision medicine
People who have the same chronic disease diagnosis or symptoms may have different mechanistic causes and will therefore benefit from different care pathways.
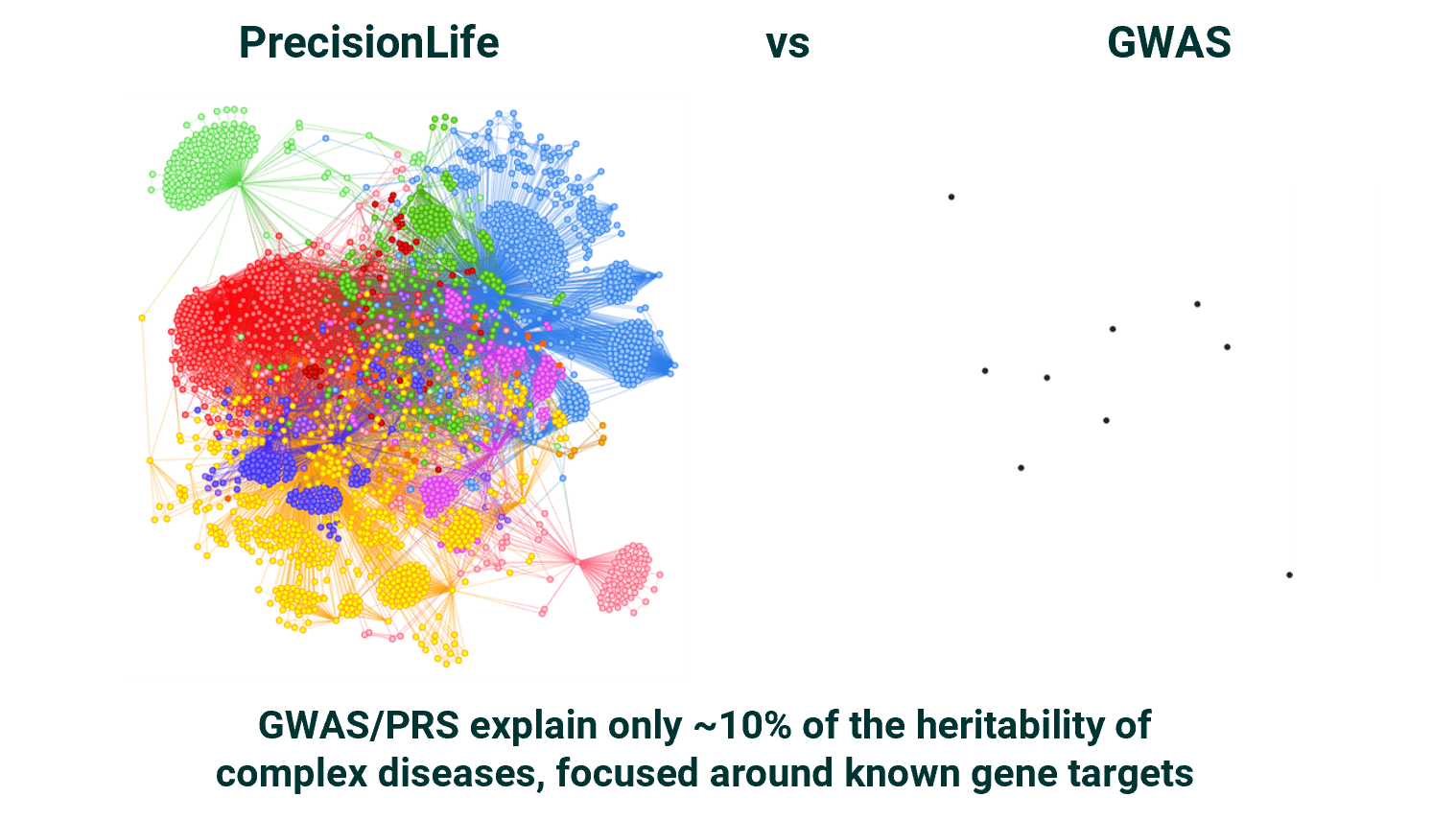
These mechanistic differences a too subtle to be found by analysis tools such as GWAS, AI, large language models, polygenic risk scores (PRS) or knowledge graphs, which are inherently limited, lacking in patient context, and containing annotation biases.
We go far beyond these methods, to find rich signal where others find none and explainable mechanistic insights to stratify patient populations from which more personalized decisions can be made.
Finding 10x signal from 10% of the data
Predictive disease models
With unique insights into the mechanisms driving complex diseases, we discover the biomarkers that reveal which individuals share that underlying biology.
This enables us to identify predictive risk signatures, uncover novel drug targets for specific patients, and enrich and de-risk clinical trials.
We help diagnose patients more accurately and, as new programs move into the clinic, match them to the treatments most likely to work – improving outcomes and reducing the cost of care.
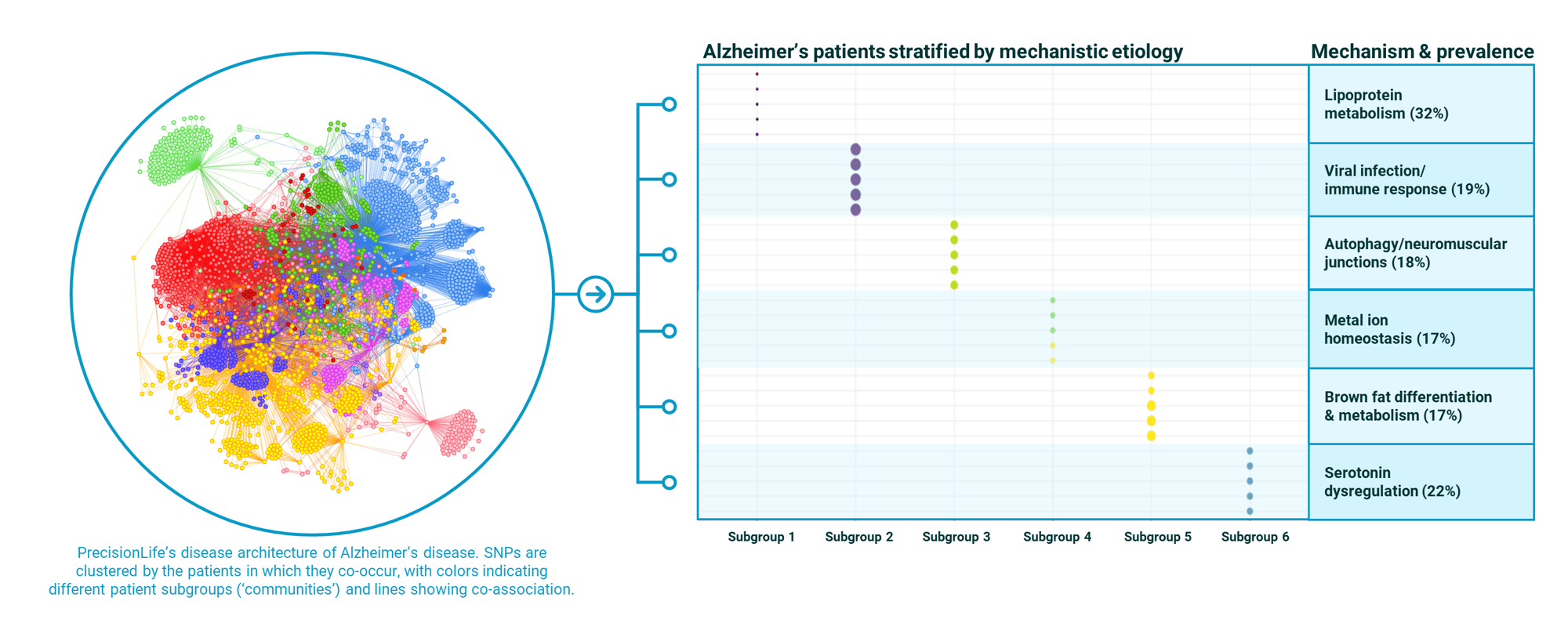
An example is our Alzheimer’s disease study, which stratified an Alzheimer's patient population into six distinct subgroups, each with a different mechanistic etiology and high prevalence.
Individuals in each group will respond to different therapies. Our mechanistic patient stratification biomarkers predict this response to optimize outcomes in drug discovery, clinical trials and healthcare.
This level of stratification across complex diseases is unique to PrecisionLife.
.png?width=683&height=683&name=Actionable%20insights%20to%20clinical%20practice-min%20(3).png)
Actionable insights to clinical practice
Our biomarkers are rapidly reduced to practice in the form of mechanostics - first-in-class, low-cost and non-invasive genotypic precision medicine tests.
Mechanostic tests identify lifetime disease risk and the underlying mechanism of an individual's disease.
The results support earlier and more accurate disease diagnosis, personalized therapy selection, and clinical trials enriched with patients who will respond to treatment.
Disease coverage
Unparalleled scalability across hundreds of complex diseases and endotypes, including:
Mental health
Anxiety panic disorder
Bipolar disorder
Obsessive compulsive disorder
Anxiety panic disorder
Schizophrenia
SSRI response
Allergy/Infectious
Acute respiratory distress syndrome
COVID-19
Fibromyalgia
Long COVID
Myalgic encephalomyelitis (ME/CFS)
Sepsis
Metabolic
Type-I diabetes
Type-II diabetes complications
Chronic kidney disease
MASH/MASLD
Obesity
Neurology
ALS/MND
Alzheimer's disease
Epilepsy
Frontotemporal dementia
Lewy body dementia
Migraine
Multiple sclerosis
Parkinson's disease
Aging
Chronic disease onset / progression
Glaucoma
Longevity
Macular degeneration
Osteoarthritis
Respiratory
Asthma (T2 and non-T2)
Bronchiectasis
Chronic obstructive pulmonary disease
Idiopathic pulmonary fibrosis
Women's health
Adenomyosis
Endometriosis
Polycystic ovary syndrome
Post-partum depression
Uterine fibroids
Cardiovascular
Atherosclerosis
Atrial fibrillation
Cardiomyopathy
Coronary artery disease
Heart failure
Hypertension
Auto-immune
Inflammatory bowel disease
Rheumatoid arthritis
Sjögren’s disease
Systemic lupus erythematosus
Systemic sclerosis
Improving health, for everyone
Our approach reduces the cost of providing care while improving outcomes for patients by identifying those who would benefit from specific therapies and clinical trials earlier - offering evidence-led access to effective medicines and a route to improved health equity.







